
TESTED
SHIPPING
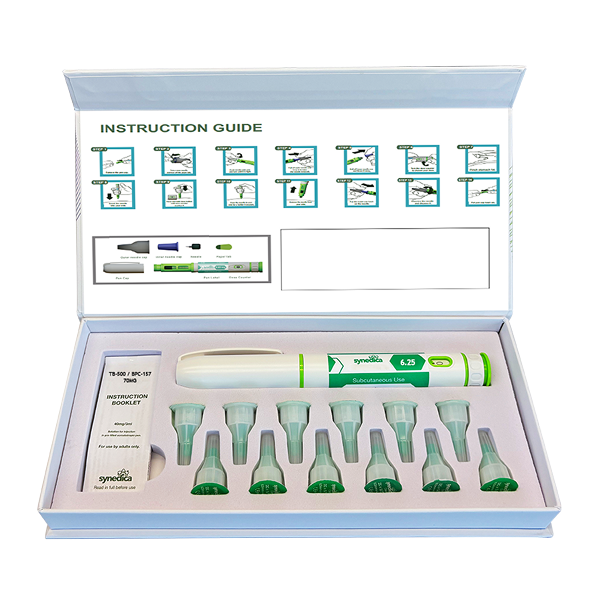
GLOW GHK-Cu / TB-500 / BPC-157 70mg Injection Dual Pen Kit
Synedica GLOW® Injection Pen Kit is an advanced regenerative peptide formulation designed to support skin rejuvenation, hair vitality, and cellular renewal.
GHK-Cu is widely recognised for its role in supporting collagen production, improving skin elasticity, and promoting tissue repair. It contributes to healthier looking skin and supports hair follicle function by encouraging cellular renewal.
TB-500 supports tissue repair and recovery by promoting cell migration, blood vessel formation, and improved circulation to damaged areas.
BPC-157 is known for its regenerative properties. It supports tissue repair and recovery by promoting angiogenesis (formation of new blood vessels) and supporting faster healing processes.
Synedica GLOW® Injection Pen Kit contains 1 pen, 12 disposable needles and an instruction booklet.
71.4% GHK-Cu, 14.3% TB-500 and 14.3% BPC-157.
Each 3ml pen contains 35mg which can be delivered across 4 full doses of 8.75mg, or in increments of 1.75mg, 3.5mg, 5.25mg, 7mg or 8.75mg total active content.
1.25mg GHK-Cu, 0.25mg TB-500, 0.25mg BPC-157 2.5mg GHK-Cu, 0.5mg TB-500, 0.5mg BPC-157 3.75mg GHK-Cu, 0.75mg TB-500, 0.75mg BPC-157 5mg GHK-Cu, 1mg TB-500, 1mg BPC-157 6.25mg GHK-Cu, 1.25mg TB-500, 1.25mg BPC-157 Recommended dosage is 1.25mg GHK-Cu, 0.25mg TB-500 and 0.25mg BPC-157 taken once daily.
All products are produced exclusively for controlled laboratory research and development applications.
Not for human or animal consumption.
Suitable R&D Applications
Stability Testing
- •Thermal, enzymatic, and oxidative degradation studies
- •Shelf-life evaluation & formulation robustness
Formulation Development
- •Enhances solubility, bioavailability, and structural integrity
- •Tailored for injectable, topical, and research-grade use
Excipient Compatibility Testing
- •Enhances solubility, bioavailability, and structural integrity
- •Tailored for injectable, topical, and research-grade use
Protease Resistance Evaluation
- •Evaluates interactions between peptides & inactive components
- •Ensures safety, stability, and performance
Encapsulation & Carrier Systems
- •Liposomes, microspheres, and other delivery vehicles
- •Protects peptides & enables controlled release
Administration Route Assessment
- •Parenteral delivery (injectables)
- •Dermal absorption (cosmetic/topical)
- •In vitro research applications